European ERA-Net Consortium on single cell Transcriptomics in CSF
Diseases named “Autoimmune Encephalitis” (AIE) are increasingly recognized as immune-mediated disorder of the gray matter (i.e. the neurons) of the central nervous system (CNS). AIE is rare, but often severely disabling and potentially lethal. In this proposal we aim to study why the immune system erroneously targets auto-immunity against neurons and synapses by integrating data from multiple novel transcriptomic and proteomic techniques.
We previously found a small number of markers that are predictive for the disease, but a full-scale unbiased characterization of autoimmunity in AIE has not been performed. We here propose to characterize the cell types, markers, pathways and genes that are specific to AIE with extremely high resolution and precision. We are a unique and focused group of clinicians and researchers from multiple countries (The Netherlands, Germany, Israel) and multiple centres (Rotterdam, Münster, Kiel, Rechovot) treating and studying a large cohort of AIE patients. We also manage large biomaterial banks. We have also developed and calibrated new genomic technologies to construct very detailed models of immune cells and pathways in autoimmune diseases. Our group thus combines cutting-edge technology with access to rare clinical specimen and a long-standing expertise in diagnosing this heterogeneous disorder. This will enable us to better understand how immune cells interact with the inflamed brain.
This outstanding combination of world leading scientists and clinicians with new technologies makes us especially well equipped to break new grounds in AIE research. We propose that an ultra-high resolution phenotyping of local immune responses, using single cell genomic analysis of the CSF will provide a basis for better understanding, diagnosing, predicting and treating AIE in the future.
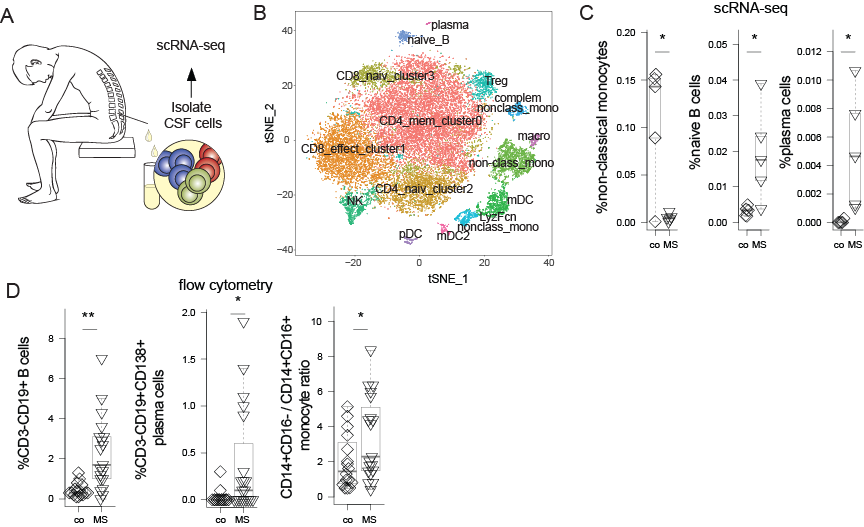
Figure 1: Illustration of diagnostic workflow in scRNA-seq of CSF cells, that identifies MS-specific changes. (A) CSF cells were collected form 5 controls and 5 multiple sclerosis (MS) patients. (B) Dimensionality reduction using tSNE was performed. Modified from PubMed ID 31937773.
Contact:
PD Dr. med. Gerd Meyer zu Hörste
Oberarzt und Arbeitsgruppenleiter
Klinik für Neurologie mit
Institut für Translationale Neurologie
Universitätsklinikum Münster
Albert-Schweitzer-Campus 1, Gebäude A1
48149 Münster
E-mail: gerd.mzh@uni-muenster.de
medizin.uni-muenster.de/en/mzhlab
Prof. Dr. Maarten Titulaer
Neurologist
Erasmus University
Department of Neurology
s-Gravendijkwal 230
3015 CE Rotterdam, Netherlands
E-mail: m.titulaer@erasmusmc.nl
https://www.erasmusmc.nl/nl-nl/patientenzorg/specialismen/neurologie-volw
Prof. Ido Amit
Principal Investigator
Weizmann Institute of Science
Department of Immunology
Wolfson Building for Biological Research
Rehovot, 76100, Israel
E-mail: ido.amit@weizmann.ac.il
https://www.weizmann.ac.il/immunology/AmitLab/ido-amit-lab-homepage
Prof. Dr. Frank Leypoldt
Facharzt für Neurologie, Facharzt für Laboratoriumsmedizin, Leitung Neuroimmunologische Ambulanz
Institut für Klinische Chemie
Universitätsklinikum Schleswig-Holstein
Arnold-Heller-Str. 3 (Haus U30)
24105 Kiel
E-mail: frank.leypoldt@uksh.de
https://www.uksh.de/index.php?id=1&lang=de&site=Klinische_Chemie
PD Dr. med. Nico Melzer
Oberarzt, Arbeitsgruppenleiter
Klinik und Poliklinik für Neurologie mit Institut für Translationale Neurologie
Westfälische Wilhelms-Universität Münster
Albert-Schweizer-Campus 1, Gebäude A1
48149 Münster
E-mail: nico.melzer@ukmuenster.de



